Corrective And Preventive Action Form. Here's what you need to know about corrective action and preventative action. Capa is fully followed by the pdca cycle for implementation of the ans: When the action plan has been finalized or discussed by the team, it is then documented using a action form. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. The nonconformity lives and needs to be addressed. Does your company have good processes in place for capa? Corrective action treats a nonconformity or problem that has already occurred. The full form of capa is corrective action & preventive action. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Preventive action determines and eliminates potential causes of non conformities. Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. Corrective and preventive action is also a part of the quality management system. This means the non conformity has not yet happen. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors.
Corrective And Preventive Action Form. Corrective And Preventive Actions Will Typically Have A Form That Is Used To Record The Details Of The Activity Performed To Satisfy The Event;
The Beginner S Guide To Capa Smartsheet. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. Here's what you need to know about corrective action and preventative action. The full form of capa is corrective action & preventive action. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors. Corrective and preventive action is also a part of the quality management system. When the action plan has been finalized or discussed by the team, it is then documented using a action form. The nonconformity lives and needs to be addressed. Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. Does your company have good processes in place for capa? Corrective action treats a nonconformity or problem that has already occurred. Capa is fully followed by the pdca cycle for implementation of the ans: Preventive action determines and eliminates potential causes of non conformities. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. This means the non conformity has not yet happen.
Describe the three terms with suitable example(s).
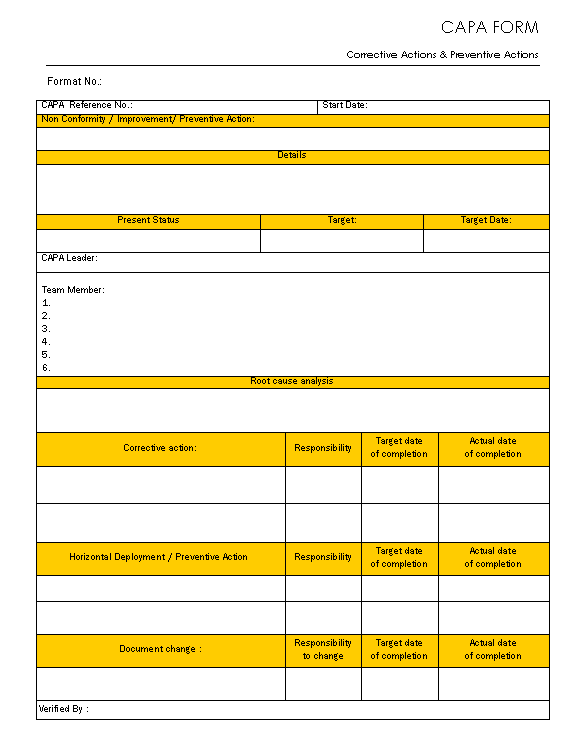
The ultimate purpose of capa is to assure the problem can never be experienced again. You must understand these terms for a however, differentiating corrective action and preventive action is a little difficult; This means the non conformity has not yet happen. In short, preventive action prevents occurrence and corrective action prevents recurrence, while correction basically refers to containment. Corrective and preventive action application. Root causes of nonconforming product 1.3 any employee of the company can initiate a corrective/ preventive action request by completing the top portion of the corrective action request (car) form, but only. Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. This type of informal and incremental. Here's what you need to know about corrective action and preventative action. Are there situations where both preventive action and corrective action are undesirable and correction is the only p. The action was taken to eliminate the cause of deviation, defect, or other undesirable situation in order to prevent the future occurrence of such or similar an e) write in brief the capa description from the source document and corrective and preventive action details. Corrective action is part of quality management and involves a set of actions taken to rectify a process, task or an employee's behavior when they are at risk of producing errors or deviating from. Corrective and preventive actions will typically have a form that is used to record the details of the activity performed to satisfy the event; Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Preventive action determines and eliminates potential causes of non conformities. A corrective action request (car, pronounced car, as in an automobile) and a preventive action request (par, pronounced like the golf term, par). Effective corrective action and preventive action (capa) systems are a key component to corrections/corrective and preventive actions for quality. F) the department head shall write. This is an important step in elaborating the steps that must be taken to change standard operating procedures in order to eliminate recurring. Capa can be applied in. Capa is fully followed by the pdca cycle for implementation of the ans: Corrective and preventative actions in the workplace. Issues, risks and nonconformity should a sample action plan form is included. The nonconformity lives and needs to be addressed. Form used for corrective action requests, opportunities for improvement and preventive action requests. Does your company have good processes in place for capa? It is usually a set of actions that laws or regulations require an organization to take in manufacturing. Eliminate the root cause and. Corrective action preventive action (capa) is a process which investigates and solves problems, identifies causes, takes corrective action and prevents recurrence of the root causes. Corrective and preventive actions are used every day, in every day situations. The difference is so minuscule that many aspirants get confused.
Preventive Action By Iso 9001 Checklist Issuu- Capa Can Be Applied In.
Correction Corrective Action And Preventive Action We Ask And You Answer The Best Answer Wins Benchmark Six Sigma Forum. The full form of capa is corrective action & preventive action. Preventive action determines and eliminates potential causes of non conformities. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. Here's what you need to know about corrective action and preventative action. When the action plan has been finalized or discussed by the team, it is then documented using a action form. This means the non conformity has not yet happen. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. Does your company have good processes in place for capa? Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. Capa is fully followed by the pdca cycle for implementation of the ans: Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Corrective action treats a nonconformity or problem that has already occurred. Corrective and preventive action is also a part of the quality management system. The nonconformity lives and needs to be addressed. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors.
Corrective And Preventive Action Capa Program Sop , Corrective And Preventive Action Application.
Correction Corrective Action And Preventive Action We Ask And You Answer The Best Answer Wins Benchmark Six Sigma Forum. Capa is fully followed by the pdca cycle for implementation of the ans: The full form of capa is corrective action & preventive action. Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. Corrective and preventive action is also a part of the quality management system. Here's what you need to know about corrective action and preventative action. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. This means the non conformity has not yet happen. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. When the action plan has been finalized or discussed by the team, it is then documented using a action form.
Corrective Preventive Action Authorstream . Capa can be applied in.
Preventive Action Report Iso Template. Capa is fully followed by the pdca cycle for implementation of the ans: Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. When the action plan has been finalized or discussed by the team, it is then documented using a action form. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Corrective and preventive action is also a part of the quality management system. Here's what you need to know about corrective action and preventative action. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. Corrective action treats a nonconformity or problem that has already occurred. Preventive action determines and eliminates potential causes of non conformities. This means the non conformity has not yet happen. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors. The nonconformity lives and needs to be addressed. Does your company have good processes in place for capa? It is usually a set of actions that laws or regulations require an organization to take in manufacturing. The full form of capa is corrective action & preventive action.
Capa When To Take Corrective Action Or Preventive Action . Here's What You Need To Know About Corrective Action And Preventative Action.
Corrective And Preventive Actions Capa Fda. The nonconformity lives and needs to be addressed. Corrective and preventive action is also a part of the quality management system. Corrective action treats a nonconformity or problem that has already occurred. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Here's what you need to know about corrective action and preventative action. Does your company have good processes in place for capa? This means the non conformity has not yet happen. Preventive action determines and eliminates potential causes of non conformities. Capa is fully followed by the pdca cycle for implementation of the ans: When the action plan has been finalized or discussed by the team, it is then documented using a action form. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. The full form of capa is corrective action & preventive action. Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries.
Capa When To Take Corrective Action Or Preventive Action . This Means The Non Conformity Has Not Yet Happen.
Iso 9001 Corrective And Preventive Action Form Beautiful Corrective Action Flow Chart Onourway Models Form Ideas. Does your company have good processes in place for capa? Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors. Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. The full form of capa is corrective action & preventive action. This means the non conformity has not yet happen. When the action plan has been finalized or discussed by the team, it is then documented using a action form. The nonconformity lives and needs to be addressed. Corrective action treats a nonconformity or problem that has already occurred. Preventive action determines and eliminates potential causes of non conformities. Here's what you need to know about corrective action and preventative action. Corrective and preventive action is also a part of the quality management system. Capa is fully followed by the pdca cycle for implementation of the ans:
Corrective Action And Preventive Action Procedure Start Ppt Download - Root Causes Of Nonconforming Product 1.3 Any Employee Of The Company Can Initiate A Corrective/ Preventive Action Request By Completing The Top Portion Of The Corrective Action Request (Car) Form, But Only.
Corrective And Preventive Action Ppt Download. Corrective action treats a nonconformity or problem that has already occurred. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Here's what you need to know about corrective action and preventative action. When the action plan has been finalized or discussed by the team, it is then documented using a action form. Corrective and preventive action is also a part of the quality management system. Capa is fully followed by the pdca cycle for implementation of the ans: The full form of capa is corrective action & preventive action. Preventive action determines and eliminates potential causes of non conformities. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors. The nonconformity lives and needs to be addressed. Does your company have good processes in place for capa? Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. This means the non conformity has not yet happen.
Corrective And Preventive Action Environmental Policy Government , Corrective And Preventive Actions Will Typically Have A Form That Is Used To Record The Details Of The Activity Performed To Satisfy The Event;
Frm 820 Capa Corrective Preventive Action Med Dev Form. Capa is fully followed by the pdca cycle for implementation of the ans: The full form of capa is corrective action & preventive action. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. Here's what you need to know about corrective action and preventative action. Does your company have good processes in place for capa? Preventive action determines and eliminates potential causes of non conformities. The nonconformity lives and needs to be addressed. Corrective action treats a nonconformity or problem that has already occurred. Corrective and preventive action is also a part of the quality management system. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors. When the action plan has been finalized or discussed by the team, it is then documented using a action form. This means the non conformity has not yet happen. Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations.
Corrective Action Forms Implementation And Measurement Tips , Corrective And Preventive Action Plans Are Formulated In Order To Improve The Organizations Processes To Eliminate Causes Of Incidents Or Undesirable Situations.
Difference Between Corrective Action And Preventive Action Difference Between. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Capa is fully followed by the pdca cycle for implementation of the ans: Does your company have good processes in place for capa? Preventive action determines and eliminates potential causes of non conformities. Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. Corrective action treats a nonconformity or problem that has already occurred. The full form of capa is corrective action & preventive action. When the action plan has been finalized or discussed by the team, it is then documented using a action form. This means the non conformity has not yet happen. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors. The nonconformity lives and needs to be addressed. Here's what you need to know about corrective action and preventative action. Corrective and preventive action is also a part of the quality management system. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations.
Capa Beginner S Guide To Corrective Action Preventive Action , F) The Department Head Shall Write.
Correction Corrective Action And Preventive Action We Ask And You Answer The Best Answer Wins Benchmark Six Sigma Forum. The nonconformity lives and needs to be addressed. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors. The full form of capa is corrective action & preventive action. It is usually a set of actions that laws or regulations require an organization to take in manufacturing. Here's what you need to know about corrective action and preventative action. When the action plan has been finalized or discussed by the team, it is then documented using a action form. Capa is fully followed by the pdca cycle for implementation of the ans: Preventive action determines and eliminates potential causes of non conformities. This means the non conformity has not yet happen. Corrective action treats a nonconformity or problem that has already occurred. Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. Corrective and preventive action is also a part of the quality management system. Does your company have good processes in place for capa? Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations.
Corrective And Preventive Action Procedure - Both Preventive And Corrective Actions Are Developed To Improve An Organization's Management Systems, And Their Main Difference Can Be Identified By Taking A Closer Look At Their Definition Which Is Found In Most Of The Iso Standards.
Corrective And Preventive Action Example Usable Examples. Corrective action treats a nonconformity or problem that has already occurred. When the action plan has been finalized or discussed by the team, it is then documented using a action form. This means the non conformity has not yet happen. Preventive action determines and eliminates potential causes of non conformities. Does your company have good processes in place for capa? Capa is fully followed by the pdca cycle for implementation of the ans: Learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. Corrective and preventive action is also considered a tool within six sigma for understanding regular business operations. Mastercontrol corrective action and preventive action forms help management alter existing procedural methods to eliminate the occurrence of errors. The full form of capa is corrective action & preventive action. Here's what you need to know about corrective action and preventative action. The nonconformity lives and needs to be addressed. Corrective and preventive action is also a part of the quality management system. Corrective and preventive action plans are formulated in order to improve the organizations processes to eliminate causes of incidents or undesirable situations. It is usually a set of actions that laws or regulations require an organization to take in manufacturing.